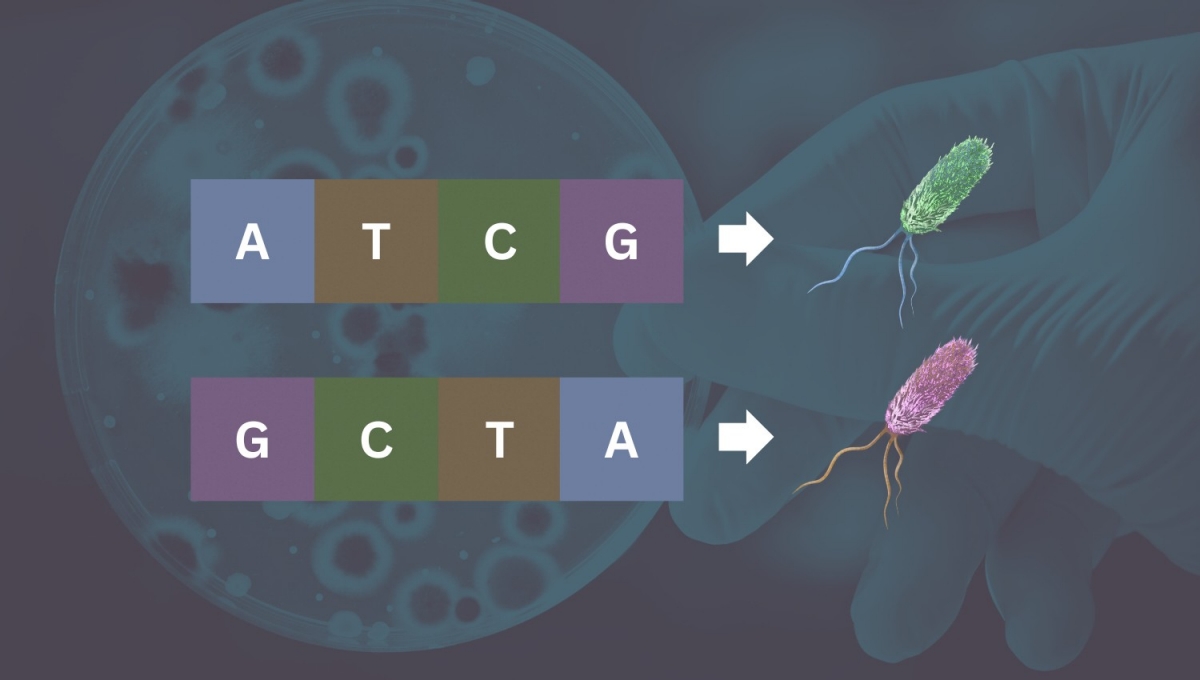
Researchers have found that inversions, which cause a physical flip of a segment of DNA and change an organism’s genetic identity, can occur within a single gene. Emily Moskal and Alexander Raths - stock.adobe.com
Imagine being one cartwheel away from changing your appearance. One flip, and your brunette locks are platinum blond. That’s not too far from what happens in some prokaryotes, or single-cell organisms, such as bacteria, that undergo something called inversions.
A study led by scientists at Stanford Medicine has shown that inversions, which cause a physical flip of a segment of DNA and change an organism’s genetic identity, can occur within a single gene, challenging a central dogma of biology — that one gene can code for only one protein.
“Bacteria are even cooler than I originally thought, and I’m a microbiologist, so I already thought they were pretty cool,” said Rachael Chanin, PhD, a postdoctoral scholar in hematology. Microbiologists have known for decades that bacteria can flip small sections of their DNA to activate or deactivate genes, Chanin said. To the team’s knowledge, however, those somersaulting pieces have never been found within the confines of a single gene.
In the same way that reversing the order of the letters in the word “dog” could entirely change the meaning of a sentence (“I’m a dog.” versus “I’m a god.”), the within-gene inversion essentially recodes the bacterium’s genetics using the same material. That could result in the activation of a gene, a halt in gene activity or a sequence that codes for the creation of a different protein when inverted.
“I remember seeing the data, and I thought, ‘No way, this can’t be right, because it’s too crazy to be true,’” said Ami Bhatt, PhD, professor of genetics and of medicine. “We then spent the next several years trying to convince ourselves that we had made a mistake. But as far as we can tell, we have not.”
A study detailing the scientists’ findings was published Sept. 25 in Nature. Chanin and former postdoctoral scholar Patrick West, PhD, co-led the study. Bhatt is the senior author.
Flip-flopping
In the 1920s, scientists encountered the first hint of inversions when they were searching for a salmonella treatment. They tried collecting antibodies from animals infected by the bacteria in the hopes that the immune molecules could be transferred to other animals and stave off infection. But it never worked — even the bacterial strains they knew to be genetically identical were able to fight back. Scientists now know that evasion was thanks to an inversion recoding the bacterium in a way that allowed it to escape the animals’ immunity.
Microbiologists have since found inversions occurring in small DNA segments of various kinds of prokaryotes. But Bhatt and her team wondered if they could also happen within a single gene. West created an algorithm, called PhaVa, that identified possible inversions within bacterial genomes.
The software essentially downloads thousands of genome sequence segments from various prokaryotes and scans for regions that look “flippable” — segments with something called inverted repeats, which have a redundant palindromic quality (for instance, ATTCC and CCTTA) — on other side of the potential inversion. The algorithm creates a catalog of what these sequences would look like if they flipped, and it makes comparisons between the made-up genomes and the true sequence. Then it counts the regions where both the flipped and unflipped sequences are present within an organism’s genome, with every match indicating a likely inversion.
The software identified thousands of inversions that exist in bacterial and other prokaryotic species, revealing for the first time that inversions occur within genes. That sparked the idea that not only do single-gene inversions occur, but that they may be relatively common, Bhatt said.
“This was really surprising to us,” Bhatt said. “To our knowledge, this has never been seen before.”
One big question remains: What causes an inversion? The team suspects there are specific enzymes that mediate the flip, as well as certain environmental cues that drive the change.
“That’s a to-do now,” Bhatt said. “One of our next steps is to try to decode the molecular grammar so we can build a database of enzymes and a database of the inverted repeats that they flip.”
Interpreting inversions
While there’s still much more to understand about inversions, Bhatt sees potential for numerous applications. “This is effectively a heritable, reversible type of genetic regulation,” she said.
She posits that scientists may eventually be able to use inversions to create a toggleable bacterial system to control their gene expression, something that could behoove synthetic biology research. Or perhaps there are links between certain diseases and the state of bacterial inversions, in which case there may be a way to switch the bacteria’s state and regulate a disease.
“This type of adaptation has just been hiding in front of us, waiting for the right tool and the right technology and biological question to be asked,” Chanin said. “And it makes me wonder, how many more bacterial secrets are just waiting for us to uncover them?”
Researchers from Princeton University contributed to this study.
This study was funded by the National Institutes of Health (grants R01 AI148623, R01 AI143757, R01 AI174515, HG000044, HL120824, TL1TR003019, 1S10OD02014101), the AP Giannini Foundation, the National Science Foundation Graduate Research Fellowship, the Stanford DARE fellowship and the Stand Up 2 Cancer Foundation.