8/7/96
CONTACT: David F. Salisbury, News Service (650) 723-2558;
e-mail: david.salisbury@forsythe.stanford.edu
Background
Zare lab method for detecting organic materials in meteorites
STANFORD -- Chemists have a number of ways to identify small amounts of organic compounds within materials. These generally involve grinding up a small sample, dousing it with various chemicals, separating the mixture by some means, and recording the results.
Such methods, however, have several major limitations. They have difficulty detecting trace amounts of molecules; they often require large amounts of the sample for processing; and they destroy the material involved.
In the last decade, however, a laser technique developed by Richard N. Zare, professor of chemistry at Stanford University, has provided an alternative method for measuring such compounds that does not suffer from these limitations. Called Microprobe Two-Step Laser Microscopy, it measures a wide range of organic compounds directly, without altering or preparing the sample or requiring an intervening separation step. It is extremely sensitive, and is capable of detecting the presence of species down to trace levels. It can simultaneously determine the relative abundance of a number of different organic compounds, but is restricted to only certain classes of these compounds.
The method was initially applied to biomolecules including amino acids and simple peptides in order to help determine protein structure, but it has proven particularly effective in measuring a certain class of organic molecules in meteorites called polycyclic aromatic hydrocarbons. PAHs consist of fused, six-member rings. They are found in coal and incomplete combustion of organic compounds, such as diesel exhaust or steak charred on the barbecue.
The Zare lab has used its probe for detecting the first organic molecules of both an interplanetary and interstellar origin in individual grains within meteorites. Most recently the method has been used to find organic material in a meteorite of Martian origin.
Zare got the idea for this new kind of instrument in 1987 when he heard about work being done in Germany that used a laser to detach molecules from a surface and then swept them into a detector using a supersonic jet of gas.
"I saw several problems with the technique and thought that I knew a better approach," Zare said.
The basic idea is very simple.
First, an infrared laser pulse heats a small area, about size of the dot at the end of this sentence. The laser beam has a tremendous power density, more than a megawatt per square centimeter. It causes the surface to heat rapidly, at a rate of more than 100 million degrees per second. But the pulse is very short, lasting only 10 millionths of a second. Such a rapid and intense blast of heat has proven to be a very effective way to detach organic molecules from a solid surface without damaging either the molecules or the sample.
"Nobody understands why this works as well as it does," Zare said.
Organic molecules are not the only ones vaporized by the first laser pulse. A lot of other molecules stuck to the surface are "desorbed" as well. So the next step is to blast the vapor cloud with an intense pulse of ultraviolet laser light. The second beam carries an even greater punch, with a power density of 60 megawatts per square centimeter, but it lasts for even a shorter time, a billionth of a second. This beam first excites certain types of molecules and then knocks an electron off, leaving them with a positive electric charge.
All this is done in a very high vacuum and electric field. As soon as the molecules become electrically charged, they are impelled out of the vapor cloud and toward a special kind of instrument called a time-of-flight mass spectrometer. Because heavier molecules move more slowly than lighter molecules, the mass spectrometer can determine their mass by clocking the time it takes them to reach the detector. On average, these molecules take about 65 microseconds to travel the three meters to the detector and so must travel at something greater than 100,000 meters per second.
The ultraviolet laser can be tuned, its frequency adjusted to pick out certain families of organic molecules. At one setting, for example, it ionizes polycyclic aromatic hydrocarbons. At another setting, it ionizes amino acids. At a third setting, it selects DNA bases. This selectivity, combined with the weight determination made by the mass spectrometer, allow the scientists to identify the relative abundance of a large number of different organic compounds with unprecedented accuracy.
One of the biggest practical problems in making the technique work was making sure that "we weren't sampling ourselves," as Zare puts it. The technique is so sensitive that it is very difficult to ensure that the organic materials being measured came from the sample and not the instrument itself. Minute amounts of pump oil, fingerprints and other organic contaminants can produce false detections. After a considerable amount of effort, which has extended over the course of four Ph.D. theses, Zare and his graduate students developed techniques that eliminate all potential sources of contamination.
Since Zare developed the technique (Stanford University holds related patents), it has been duplicated in several other university laboratories around the world, mostly by former students. For example, Renato Zenobi, now a professor at ETH University in Zurich, is using the technique to study the composition of polymer materials, and Pat Langrage-Smith at the University of Edinborough has adapted it to the study of soil pollution.
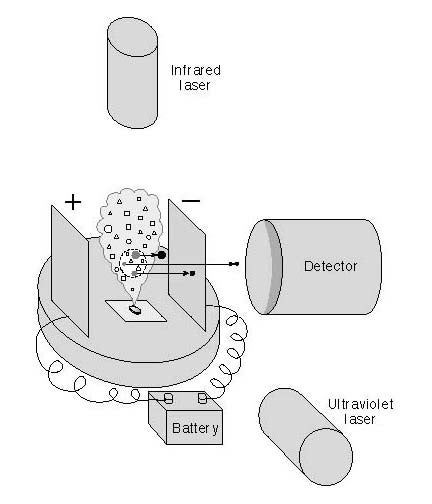
How microprobe two-step laser microscopy works to detect organic compounds:
Step 1: The pulsed infrared laser heats a tiny sample of meteorite, the size of the period at the end of this sentence. Molecules vaporize from the surface and spread in a plume within the vacuum chamber.
Step 2: The second laser, a pulsed ultraviolet laser, ionizes some of the molecules in the plume. For this experiment, the UV laser was tuned to ionize only a family of molecules called polycyclic aromatic hydrocarbons, or PAHs. Other molecules are unaffected.
Step 3: Ionization gives the particles a positive charge: They flow in an electric field toward a detector, which can distinguish different types of PAHs from one another by their speed of travel heavy PAHs travel more slowly than light ones.
-dfs-
960807marsprobe.html
Download this release and its related files.
The release is provided in Adobe Acrobat format. Any images shown in the release are provided at publishing quality. Additional images also may be provided. Complete credit and caption information is included.
960807marsprobe.sit
